Newest
-

Leading the Trillion-Dollar Agentic Commerce Race: Tec-Do Anchors Its Position in the $100 Billion MarTech Ecosystem
-

Inside Korea’s intense exam season: From grounded flights to police escorts to school
-

Bodycams found on divers killed in Maldives could reveal final moments
-

Man dies ‘while being pinned down by shop security for shoplifting’
Lunit to Present AI-Analyzed Immune Phenotype Study as Immunotherapy Response Predictor for Advanced Gastric Cancer at ESMO Congress 2024

Lunit SCOPE IO® demonstrates potential to assess immune phenotype as an AI-powered biomarker for predicting efficacy of Nivolumab plus Chemotherapy in advanced gastric cancer,independent of PD-L1 status
SEOUL,South Korea,Sept. 10,2024 -- Lunit (KRX:328130.KQ),a leading provider of AI-powered solutions for cancer diagnostics and therapeutics,today announced the presentation of a significant study at the European Society for Medical Oncology (ESMO) Congress 2024 in Barcelona,Spain,from September 13-17.
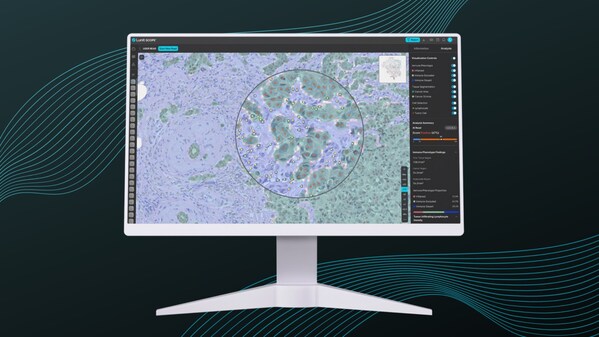
Lunit's AI-powered histopathology analyzer,Lunit SCOPE IO
While Nivolumab plus chemotherapy has recently been approved as a standard first-line treatment for advanced gastric cancer (AGC),its efficacy varies among patients. This variability underscores the critical need for reliable biomarkers to predict treatment response. Lunit's study addresses this pressing need,potentially offering a new tool to optimize patient care and treatment decisions.
Conducted in collaboration with leading Korean medical institutions,the research showcases the potential of Lunit's AI-powered histopathology analyzer,Lunit SCOPE IO®,in predicting treatment response and guiding treatment decisions for AGC via assessment of immune phenotype.
The study analyzed H&E images from 585 AGC patients,with Lunit SCOPE IO® classifying tumors into two immune phenotypes—inflamed (IIP) and non-inflamed—based on the presence and distribution of tumor-infiltrating lymphocytes (TILs) from hematoxylin and eosin (H&E) slides that are readily available from a standard clinical workup. This classification provided valuable insights into the tumor microenvironment and helped predict treatment response,particularly in cases where traditional biomarkers like PD-L1 may not provide a complete picture.
Key findings include:
1. Patients treated withNivolumab+Chemotherapy showed significantly longer median progression-free survival (mPFS) compared to Chemotherapy alone (8.2 vs. 5.9 months).
2. The inflamed immune phenotype group,classified byLunit SCOPE IO®,was associated with more pronounced PFS benefits from Nivolumab+Chemotherapy:
IIP: 5.2 months longer PFS for Niv+Chemo (mPFS of 11.0 vs 5.8 months)
Non-IIP: only 1.4 months longer PFS for Niv+Chemo (mPFS of 7.3 vs 5.9 months)
3. The predictive value ofIIP was consistent across different PD-L1 expression levels.
4. Multivariate analysis confirmedIIP as an independent factor for PFS in patients treated with Nivolumab+Chemotherapy.
"By demonstrating that our AI-powered immune phenotype analysis can predict treatment response independently of PD-L1 status,we're opening new possibilities for tailoring treatments in AGC," said Brandon Suh,CEO of Lunit. "This is particularly significant because gastric cancer continues to be a leading cause of cancer-related deaths globally,representing 7.7% of all cancer cases. Our AI technology has the potential to enhance the precision of treatment decisions,potentially leading to more effective therapies and better quality of life for patients with AGC."
Please visit Lunit's poster session at 1411P to discover more about our findings and the innovative capabilities of Lunit SCOPE IO®.
Poster presentation featuring Lunit SCOPE IO at ESMO Congress 2024:
"AI-powered immune phenotype predicts favorable outcomes of nivolumab (niv) plus chemotherapy (chemo) in advanced gastric cancer (AGC): A multi-center real-world data analysis" (1411P,September 16,12:00~13:00 PM)
###
About Lunit
Founded in 2013,Lunit (KRX:328130.KQ) is a medical AI company on a mission to conquer cancer. We harness AI-powered medical image analytics and AI biomarkers to ensure accurate diagnosis and optimal treatment for each cancer patient. Our FDA-cleared Lunit INSIGHT suite for cancer screening serves over 3,500 hospitals and medical institutions across 50+ countries.
Our clinical studies have been published in top journals,including the Journal of Clinical Oncology and the Lancet Digital Health,and presented at global conferences such as the ASCO and RSNA. In 2024,Lunit acquired Volpara Health Technologies,setting the stage for unparalleled synergy and accuracy,particularly in breast health and screening technologies. Headquartered in Seoul,with a network of offices worldwide,Lunit leads the global fight against cancer. Discover more at lunit.io.

 2024-09-11
2024-09-11